Notes on chemical kinetics for intermediate and B.Sc.
The most important part of geting success in any branch of science is to get a quality notes on any topic. In fact, Notes are not mere the duplicate copy of any books, rather they are the systematic arrangements of various important concepts, and problem solving to feed the understanding.
For your best result we are also providing notes on Thermodynamics, Periodic table, Gaseous state, Phase rule, Industrial chemicals and environment,
and Solid state.
Now you scroll down for Notes on Chemical Kinetics.
What happens in a chemical reaction?
In a chemical reaction,
some or all of the bonds of the reacting species are broken and new bond(s) are
formed between different atoms of the same reacting species to form the
products. Thus depending upon the nature of bonds of the reactants (whether
strong or weak), a reaction may be faster or slower. Clearly, if the bonds of
the reacting species are strong, then higher amount of energy is required to break
those bonds and the reaction becomes slower as compared to a reaction in which
the bonds of the reacting species are weak. In this regard, the
fastness or slowness of a chemical reaction is mathematically termed as the
rate of the chemical reaction.
Chemical
kinetics is the branch of chemistry which deals with the study of the type, the
rate, the factors affecting the rate and the mechanism (molecular events
occurring during the chemical reaction) of a chemical reaction.
Rate of reaction (ROR): The change in concentration (M or mol per litre) of any
reactant or product per unit time, per unit stoichiometric coefficient in a
chemical reaction is called the rate of reaction.
Mathematically,
(ROR) = Change in concentration of
any reactant or product/(Change in time . 𝛎)
Where 𝛎 is the stoichiometric coefficient.
Thus the unit of rate of reaction will
be M/sec
or mol L^-1 sec^-1
or mol L-1 min -1
For any gaseous reaction:
We have, PV =
nRT
=> P = (n/V) RT = CRT = [Gas]RT
Thus if temperature is kept constant,
[Gas] α partial pressure of the gas
This implies that, in a gaseous
reaction the rate can be expressed in terms of partial pressure of gasses.
ROR = Change in partial pressure of any
reactant or product / (time taken X stoichiometric coefficient of concerned
reactant or product)
Accordingly, the unit of rate
of reaction will be atm/sec
or atm/min
or bar/sec
or Pascal/sec etc.
Based on the consideration of time
interval we can have two different rates of reaction:
Average rate of reaction: The rate of reaction measured
and calculated for a finite time interval (finite
time interval, Δt= t2 – t1) is called the average rate of reaction.
Consider a reaction, A(g) → B(g) in which the concentrations of A and B
changes with time as follows:
|
Time in
minutes |
A (in
mol L-1) |
B (in mol
L-1) |
|
0 |
1.5 |
0 |
|
5 |
1.35 |
0.15 |
|
10 |
1.25 |
0.25 |
The
stoichiometric coefficients of both A and B in the above reaction are one.
Thus if we consider the concentrations of A and
B at time 5 minutes and 10 minutes, we have,
ROR(avg)= – Δ[A] / Δt
= Δ[B] / Δt.
= –(1.25 – 1.35) / (10 –5)
= (0.25 – 0.15) / (10 –5)
= 0.02 mol L^-1 min^ –1
Note: A negative sign is multiplied while calculating rate of reaction w.r.t. the reactant to get a
positive value because rate of reaction can’t be negative
also we can say the negative sign indicates a decrease in concentration.
Consider another reaction,
2A → 3B, for which the change in concentration during the course of reaction is as follows:
|
|
A |
B |
|
At t = 0 |
a M |
0 |
|
At any time t |
(a – 2x) M |
3x M |
The Stoichiometric
coefficients for A and B (νA and νB)
in the above reaction are 2 and 3 respectively. Thus,
ROR = – Δ[A] / (Δt. νA)
= Δ[B]
/ (Δt. νB)
= – [(a – 2x) - a] M / (t – 0) min . 2
= [3x – 0] M / (t – 0) min . 3
Instantaneous rate of reaction: Rate of reaction measured at a particular instant of
The Stoichiometric
coefficients for A and B (νA and νB)
in the above reaction are 2 and 3 respectively. Thus,
ROR
= – Δ[A] / (Δt. νA)
= Δ[B] / (Δt. νB)
= – [(a – 2x) - a] M / (t – 0) min . 2
= [3x – 0] M / (t – 0) min . 3
Instantaneous rate of reaction: Rate of reaction measured at a particular instant of
time is called the
instantaneous rate of reaction. The average rate of reaction becomes equal to instantaneous rate
of reaction when the time range (Δt) tends to zero.
=>Instantaneous rate of reaction = d[A] / (dt
. ν),
where d[A] and dt are instantaneous (infinitesimal)
change in concentration of reactant and time respectively.
Instantaneous rate of reaction can be determined graphically. Slope of the tangent at any
time t gives the instantaneous rate of reaction.
For a general reaction A → B in which the concentrations of A changes with time as given
in the graph:
From the graph,
tan (180 – θ) = – tan θ = – slope
= – (OA/OB)
=> ROR (inst)
= – d[R] / dt.1
= – (0.0 – 0.9) / (20 – 0)
= 0.045 mol L^-1 min^–1
Similarly the instantaneous rate of
reaction w.r.t. a product can be obtained from the positive slope of the
curve.
=> Instantaneous rate of
reaction
= ROR (inst) = + d[P] / (dt
. ν)
Rate of appearance, Rate of disappearance and rate of reaction:
The rate of disappearance of a reactant is defined as the
decrease in concentration of the reactant per unit time.
ROD = Decrease in concentration of the
reactant / time taken
The rate of appearance of a product is defined as the
increase in concentration of the product per unit time.
ROA = Increase in concentration of the
product / time taken
The rate of appearance, disappearance
and rate of reaction are all identical for a reaction having same stoichiometric coefficient of all the
reactants and products. e.g., A + B → C + D
But for a reaction of the type
aA + bB → cC + dD
where the stoichiometric coefficients are not the same, the rate of appearance, disappearance and rate of reaction are not identical.
In this case
the rate of reaction
= –1/a (Δ[A] / Δt)
= –1/b (Δ[B] / Δt)
= 1/c (Δ[C] / Δt)
= 1/d (Δ[D] / Δt)
Where,
–(Δ[A] / Δt)
= rate of disappearance of A and
–(Δ[B] / Δt)
= rate of disappearance of B
Δ[C] / Δt = rate of appearance of C
and Δ[D]/ Δt = rate of appearance of D
Qulem – 1: For the given reaction, N2 + 3H2 —>
2NH3, the rate of appearance of ammonia is 2 X 10-4 M
S-1. Determine the rate of disappearance of N2 and H2.
[Sometimes rate of disappearance of N2 is written as rate of
reaction in terms of N2]
Solution: The rate of appearance, disappearance and rate of reaction are
related as:
–1/1 (Δ[N2] / Δt)
= –1/3 (Δ[H2] / Δt)
= 1/2 (Δ[NH3] / Δt) ……… relation 1
Given that Δ[NH3] / Δt
= ROA of ammonia = 2 X 10-4 M S-1
From relation
1,
–1/1 (Δ[N2] / Δt) = 1/2 (Δ[NH3] / Δt)
=> – Δ[N2] / Δt = ROD of N2
= 1/2 (Δ[NH3] / Δt)
= 1/2 X 2 X 10-4 M S-1
= 1 X 10-4 M S-1
Similarly,
–1/3 (Δ[H2] / Δt)
= 1/2 (Δ[NH3] / Δt)
=> – Δ[H2] / Δt = ROD of H2
= 3/2 (Δ[NH3] / Δt)
= (3/2) X 2 X 10-4 M S^-1
= 3 X 10-4 M
S^-1
Qulem – 2: In the reaction 2H2O2 —> 2H2O
+ O2, rate of formation of oxygen is 36 gram min^-1.
Determine the rate of formation of H2O and rate of disappearance of
H2O2.
Solution: The rate of formation of oxygen = 36 gram min^-1.
Since in a chemical reaction, reactants react according to their moles and also
the products are formed accordingly w.r.t. their moles, we must convert 36 gram
min^-1 into moles.
Thus, 36 gram min-1 of
O2 = 36/32
= 1.125 mol min^-1
= Δ[O2] / Δt = rate of appearance or formation of O2.
The rate of appearance, disappearance and rate of reaction are
related as:
–1/2 (Δ[H2O2] / Δt)
= 1/2 (Δ[H2O] / Δt)
= 1/1 (Δ[O2] / Δt)
Δ[H2O] / Δt
= Rate of formation (ROA) of H2O
= 2 (Δ[O2] / Δt)
= 2 X 1.125 mol min^-1
= 2.25 mol min^-1
– Δ[H2O2] / Δt = rate of disappearance (ROD)
of H2O2
= 2 (Δ[O2] / Δt)
= 2.25 mol min^-1
Qulem – 3: In the reaction 2NO(g) + Cl2 (g) —> 2NOCl
(g) being carried out in a closed vessel, the partial pressure of NO is
decreasing at a rate 160 torr/min. Calculate the rate of appearance of NOCl.
Solution: –1/2 ROD (NO) = 1/2 ROA
(NOCl) => ROA (NOCl) = 160 torr/min = 2.67 torr/sec
Qulem – 4: Balance the chemical reaction for acidic medium and
determine the ratio of rate of change of H+ ion to the rate of
change of MnO4–.
[Fe(H2O)2 (C2O4)2]^2– + MnO4^– —>
Mn^2+ + Fe^3+ + CO2
Solution: The balanced chemical equation is:
[Fe(H2O)2 (C2O4)2]^2– + MnO4^– +
8 H^+ —> Mn^2+ + Fe^3+ +
4CO2 + 6 H2O
For the above reaction,
–1/1 ROD (MnO4^–) = 1/8 ROD
(H^+)
=> ROD (H^+) / ROD
(MnO4^–)
= 8/1 = 8:1
Factors affecting the rate of chemical
reaction: Following factors are
responsible:
1.Concentration or partial
pressure of reactants: Higher is
the concentration or the partial pressure of the reactants greater is
the number of molecules of the reactants. Hence greater will be the number
of effective collisions between them which results in the increasing rate of reaction.
This has been mentioned
also in law of mass action that the rate of chemical reaction is directly
proportional to the product of concentration of reactants.
For example, a burning
object burns rapidly in a gas jar full of oxygen.
2. Temperature: We know kinetic energy increases as the temperature
increases. Thus,
higher is the temperature, greater will
be the number of collision having sufficient
kinetic energy. Hence the rate of reaction increases.
It has been found experimentally that rate of reaction increases around 2 times for
each 10 degree centigrade rise in temperature. The ratio of specific reaction rates or the ratio of the rate
constants at two different temperatures differing by 100c is called the temperature
coefficient.
Thus T.C = K (t + 100c) / K (t 0c)
For
example, copper turnings do not
react with nitric acid at temperature but on heating they react vigorously.
3. Catalyst: A positive catalyst increases the rate of reaction by providing an alternative
pathway in which the activation energy is low.
Hence a large number of molecules cross this energy barrier and give more
products.
For
example, hydrogenation of ethane gives good yield of product in presence Ni
catalysts.
C2H4 +
H2 ------> C2H6
But
a negative catalyst decreases the rate of the
reaction as it increases the activation energy.
For
example, the decomposition of H2O2 becomes slower in
presence of phosphoric acid.
4. Surface area of reactants:
When surface area of the reactants increase, large number of
molecules are exposed to the reaction conditions. Thus processes like combustion,
adsorption etc. becomes easier. Thus higher is the surface area higher is the rate
of reaction.
For example, Zn dust reacts faster with
HCl than Zn granules.
5. Nature of reactants: Reactant molecules with strong chemical bonds require higher energy
to break these bonds hence the corresponding reaction becomes slower. On the other hand reactant molecules with weak bonds decompose easily, and react faster.
A
reaction which involves less bond rearrangement will be faster than the one in
which there are more bond rearrangement.
Reaction
also becomes faster when they are in gaseous state than when they are solid
state.
6. Radiation: The light radiation of specific
wavelength is energetic enough to break the chemical bonds of certain
chemicals. Reactions carried out using photons (light radiation) are
called photochemical reactions. Thus radiations also increase the rate of reactions.
For
example, H2 and Cl2 react slowly in dark but rapidly in presence of light.
Rate law or rate equation and rate constant: The actual relationship between the rate of reaction,
concentration of the reactants and the exponents on the reactant concentrations determined
through experiments is called the rate law or rate equation.
The proportionality constant of the rate
equation is called the rate constant. As long as the
temperature remains constant and no catalyst is used, the rate constant remains
constant. When temperature increases the rate of reaction also increases and
accordingly the rate constant also increases. Thus rate constant is directly
proportional to the rate of reaction.
Consider
the reaction,
2NO2 + F2 —> 2NO2F,
its rate of reaction is determined experimentally and is
expressed as, r = K [NO2] [F2]. It
should be kept in mind that, the stoichiometric coefficient has nothing to do
with the rate of reaction. The exponent or power of the concentration terms (1
in both [NO2] and [F2]) are written according to the
experimental data. Here K is called the rate constant.
Order of a reaction:
The sum total of the exponents (or powers) to which the concentration terms
are raised in the rate law expression or rate equation is called the order of the reaction. The power
of the concentration term of a particular reactant in the rate law is called the order of the reaction with
respect to that reactant or partial order of a reactant.
If for
the reaction
mA + nB —> products, the rate of the reaction is given by r = k [A]m [B]n, then the overall order of reaction = m + n. Also the partial order of the reaction with respect to the reactant A is m and w.r.t. B is n.
It should be noted that, for simple or elementary reaction, which takes
place in a single step without the formation of any intermediate, the partial
order of the reaction is equal to the stoichiometric coefficient (smallest integers).
The reactions which take
place in more than one step and in which at least one intermediate is formed
are called the complex reactions. For these reactions, the partial
order may or may not be equal to the stoichiometric coefficient.
Order of reactions can be of the following types:
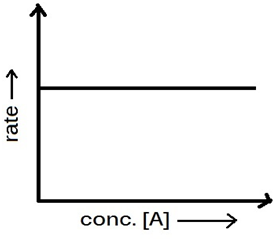
(1) Zero order reaction: The reaction in which the rate of reaction is independent upon the initial concentration of
reaction is called zero order reaction. In general, reactions taking place on the surface of the
catalysts or in presence of sunlight are zero order.
Example:1
2NH3 — > N2 + 3H2
(this reaction takes place over Pt catalyst) Rate = K [NH3]^0
2. Reaction of hydrogen and chlorine in presence of
sunlight to give hydrogen chloride is also a zero order reaction. Rate = K [H2]0 [Cl2]0
(2) First order reaction: In this type of reaction, the rate of reaction depends only upon the concentration of one
reacting species, raised to the power unity.
Example:1
PCl5 —> PCl3 +
Cl2 ;
rate = — d [PCl5]
/ dt
=> r = K [PCl5]^1
(3) Pseudo first order reaction: The reaction in which there are more than one reactants but
the rate of reaction depends only on the conc. of one
reactant species, raised to the power unity
is called pseudo first order reaction. All other reactants
being present in excess do not affect the rate of reaction.
Example: Hydrolysis of ethyl acetate with water.
CH3COOC2H5 +
H2O —> CH3COOH + C2H5OH
Rate = K [CH3COOC2H5]^1
=> Order = 1
(4) Second order reaction:
The reaction in which the rate of reaction depends upon the molar concentrations of two reacting species each
raised to the power equal to unity or the reaction in
which the rate of the reaction depends on one reacting species raised
to the power equal to two is called a second order reaction.
EX : Hydrolysis of ethyl acetate with NaOH.
CH3COOC2H5 + NaOH —> CH3COONa + C2H5OH
R = K [CH3COOC2H5]^1 [NaOH]^1 => Order of reaction = 2
(5) Fractional order reaction: The reaction in which the rate of reaction depends upon
the molar concentration of reacting species having fractional power.
Example:
CH3CHO (l) —> CH4 (g) + CO (g),
r = K [CH3CHO]^3/2 ,
order = 3/2
Characteristics of order of reaction:
i.It
depends upon the conditions of the reaction
ii.It may
be zero, whole number or a fraction.
iii.It can
be determined only experimentally
iv.The
overall order of any reaction is always positive though partial order may be
negative.
v.In case a reaction is complex and proceeds in several steps, the order of the
reaction is determined from the slowest step. (Slowest step is
the rate determining step)
Qulem – 5: If 2A + 4B —> products is an elementary
reaction, then what will be its order?
Solution: In an elementary reaction smallest possible integer of
stoichiometric coefficients are taken.
2A + 4B —> products
becomes A + 2B —> products
Thus r = K [A] [B]^2 and
order = 1 + 2 = 3
Qulem – 6: The reaction 2NO (g) + Cl2 (g) —>
2NOCl (g) has an observed rate law, r = K [NO]^2 [Cl2].
Predict whether the reaction is simple or complex.
Solution: Since the partial orders of the reaction are similar to the
stoichiometric coefficients, the reaction may be an elementary or may be a
complex reaction.
Rate constant or specific reaction rate:
Consider a reaction A + B —> Product
Rate of reaction,
r = dx /dt = K [A] [B] Where k is the proportionality constant or the rate constant
When [A] = 1 mole/ltr and
[B ] = 1 mol/ltr
then rate = K = specific
reaction rate
The rate constant (or specific reaction rate) of reaction at a given temperature is defined as
the rate of reaction when the molar conc. of each the reacting species is unity.
Units of rate constant:
Let A —> product (a first order reaction)
r = K [A] or K = r / [A]
= Mole Ltr^-1 sec^ -1 / Mole Ltr^-1
= sec^ -1
=> Unit of rate const.
of first order reaction = sec^-1
Let A —> product (a zero order reaction)
r = K [A] ^0 => K = r
=> Unit of K of
zero order reaction
= Mol lit^ -1 sec^-1
In general,
Unit of rate constant ‘K’
of nth order reaction is Mol^ (1-n) litre^(n -1) sec^ -1
Molecularity: The number of reacting species (molecules \atoms\ions)
taking part in an elementary reaction to bring about the product is called the
molecularity. This is a theoretical concept defined only for elementary
reactions.
If the decomposition of a single species brings about the product then it is a unimolecular
reaction. i.e., the number of reacting species = 1,
For Ex: NH4NO2 —> N2 + 2H2O,
or N2O5 —> N2O4 + ½ O2, The molecularity = M = 1
For any reaction of the type:
A+B —> product,
Molecularity = M =2 (called bimolecular eaction)
For Ex: CH3COOC2H5 + NaOH —> CH3COONa + C2H5 OH
For any reaction of the type:
A+B+C —>
product, or 2A + B —>
product, M = 3 (called trimolecular reaction)
For Ex: 2SO2 + O2 —> 2SO3
Characteristics of molecularity:
1. Molecularity is determined for a single step reaction or a single step of a complex reaction.
2. It is always a whole number and can’t be fraction, zero or negative.
3. The value of molecularity can’t exceed 3.
In case a reaction contains more than 3 reactants,
it can’t takes place in a single step and proceed through two or more consecutive steps.
These types of reactions are called complex reactions.
Consider a reaction:
4HBr + O2 —> 2H2O + Br2 which occurs stepwise as follows:
HBr + O2 —> HOOBr (slow)
HOOBr + HBr —> 2HOBr
HOBr + HBr —> H2O + Br2
Since the slow step contains two molecules, its molecularity is 2. Here molecularity of the
overall reaction has no significance.
Qulem – 7: You have an elementary reaction of the type
A + 2B —> Product. What happens
to the rate of reaction if volume of the vessel is reduced to one third of its
initial volume?
Solution: For an elementary reaction the partial orders are equal to
the stoichiometric coefficients.
Thus for A + 2B —> Product, the
rate, Rate1 = k [A] [B]^2
Concentration = mole/Volume. Thus when
volume is reduced to one third the concentration becomes three times.
The final Rate2 = k [3A] [3B]^2
= 27 k [A] [B]^2
Now Rate2 / Rate1 = 27. Thus the final
rate becomes 27 times faster than the initial rate.
Qulem – 8: Given a reaction
2NO (g) + Cl2 (g) —>
2NOCl (g. The rate of reaction is doubled when concentration
of Cl2 is doubled and it becomes eight times when concentration
of both NO and Cl2 are doubled. Predict the order of reaction.
Solution: Let partial
order of the reaction with respect to NO is X and w.r.t. Cl2 is
Y.
Rate = r = K [NO]^X [Cl2]^Y
Now according the question, 2r = K [NO]^X [2Cl2]^Y and 8r
= K [2NO]^X [2Cl2]^Y
Solving the above equation we get X = 2
and Y = 1, Hence the overall order of the reaction is = 2+1 = 3
Integrated rate equation:
Till now we have expressed
the concentration dependence of rate as differential rate equation. When we integrate the
differential equation, we get a direct relationship between rate of reaction, molar concentration,
rate constant and time. Such equations are called integrated rate equation.
Derivation of the rate constant of a zero order reaction
or Integrated rate equation for zero order reaction:
Consider a zero order reaction,
Rate of reaction = — d
[a — x] / dt
= dx / dt = K0 [A]^ 0
= K (a — x) ^0 = K
Where K = rate constant for zero order reaction.
Thus dx = K0 dt
Integrating both sides,
=> x = Kt => K = x/t …….. eq 1. This is called the integrated rate equation for the zero order
reaction where x is the amount of reactant consumed or the amount of product formed in time t.
If [A]0 = initial concentration of A (at t =0) and [A]t = concentration of A at any time t, then eq. 1
becomes
K = {[A]0 — [A]t} / t
=> [A]t = — Kt + [A]0 …….. eq. 2
or [A]0 — [A]t = Kt
Half life period (t1/2 or t50%):
The time in which half of the reaction is complete is called the half life period. From eq. 1, t = x / K,
when t = t1/2 then x = a/2
=> t1/2 = a / 2K, which shows that the half life period
of a zero order reaction is directly proportional to the initial concentration.
Characteristics of Zero order reaction:
1. Zero order reactions proceed with
constant rate.
2.Half life of these reactions is
directly proportional to the initial concentrations and inversely proportional
to the rate constant.
3.Rate of reaction does not change with
change in concentration of the reactant.
4.The unit of rate constant is same as
that of the rate of reaction, mol L^-1 S^-1
Derivation of the rate constant of a first
order reaction or integrated rate equation for first order reaction:
Consider a first order reaction,
Rate of reaction = dx / dt = K0 [A]^ 1
= K (a — x)1 = K (a — x)
Where K = rate constant for first order reaction.
Thus dx / (a — x) = K dt
Integrating in both sides,
∫ dx /
(a — x) = ∫ K dt
=> — ln (a – x) = Kt + C ……. Eq.1,
where C is the integration constant, whose value can be
determined as follows,
When t =
0, x = amount of reactant consumed = 0
=> —ln a = C
Putting the value of C in eq. 1, we get
— ln (a – x) = Kt — ln a
=> ln a – ln (a — x) = Kt
=> ln (a — x) = – Kt + ln a …….. eq. 2
=> ln {a / (a — x)} = Kt
=> a / (a — x) = Exp (Kt) ……. Eq. 3
=> 2.303 log {a / (a — x)} = Kt
=> (a — x) = a Exp (—
Kt) ……. Eq. 3a
=> K = (2.303 / t) log {a / (a — x)} ……. eq. 4.
This is called the integrated rate equation for the first
order reaction where x is the amount of reactant consumed or the amount of product formed in
time t.
Half life period (t1/2):
The time in which half of the reaction is complete is called the half life period.
When t = t1/2 then x = a/2, putting the value of x in eq. 3, we get,
K = (2.303 / t1/2) log {a / (a/2)} => K = (2.303 / t1/2) log 2
=> t1/2 = 0.693 / K …….. eq. 4, which shows that the half life period of a first order reaction, does not depend on the initial
concentration.
The following graphs relate various parameters of first order reaction:
Kinetics of first order
reaction in terms of partial pressure:
Consider
a general gaseous first order reaction of the type
A(g) —>
B(g) + C (g)
Let
Pi be the initial pressure in the vessel due to A only and the total pressure
at any time t be Pt and the partial pressure of A, B and C be PA, PB and
Pc.
Consider
that the decrease in partial pressure of A in time t is X atm.
Total pressure at time t,
=
Pt = pi – X + X + X
=
Pi +
X
=>
X = pt – pi
And
PA = pi – X = pi – (Pt – pi)
=
2pi – Pt
Putting
these data into first order kinetics
K = (2.303 / t) log {a / (a — x)}, we get,
K = (2.303 / t) log (pi / PA)
=
(2.303 / t) log {(pi / (2pi – Pt)}
Characteristics
of First order reaction:
1.Rate
is directly proportional to the unit power of the concentration of reactant.
2.Half
lives of these reactions are independent of the initial concentrations.
3.The
time required for the 75% completion of the reaction is twice of its half life
period.
4.
The time required for the 99.9% completion of the reaction is ten times of its
half life period.
5.The
unit of rate constant is S^—1
Degree
of completion (α) of reaction:
The
part of the reaction which is complete in time t is called the Degree of
completion (α) of reaction.
α
= =
X/A0 => X = A0α
For
any reaction,
[A]t
= amount of reactant at any time t = [A]0 — X
= [A]0 — [A]0 α
= [A]0 (1 — α)
The
term tα represents the time required for the completion of α
part of the reaction.
For
Example: The time required for the 75% completion of the reaction is
represented as t75% or t3/4.
Qulem
– 9: What is the relationship between the rate constant of a zero order
reaction and the time required for the 75% completion of the reaction?
Solution: For zero order reaction, [A]0 —
[A]t = Kt ……. eq 1
α
= 75% = 3/4
=> [A]t
= [A]0 (1 — α)
= [A]0 (1 — 3/4)
= [A]0/4 (Putting this value in equation 1)
=> [A]0 —
[A]0/4 = K t75%
=> 3/4 [A]0 = K t75%
=> t75% = 3[A]0 /4K
Similarly, t100% = [A]0 /K
= 2 t50%
Note: When the order (n) of
reaction is 0 ≤ n < 1, it proceeds to completion. All other reaction never
proceed to completion.
Qulem
– 10: Certain reaction A —> B follows zero order kinetics.
The concentration of A drops from 3M to 1.5M in 1 hour. How much time should it
take to reach at 0.25M from 0.5M?
Solution: Since concentration
of A drops from 3M to 1.5M in 1 hour, its half life = t50% = 1
hour.
=>
k = [A]0 /2 t50%
=>
K = 3/2 = 1.5 M/hour
Since,
[A]0 — [A]t =
Kt
=>
0.5 – 0.25 = 1.5 X t
=>
t = 0.167 hour
Qulem
– 11: Show that the time required for the completion of three
fourth of a first order reaction is twice of that needed for the completion of
half of the reaction.
Solution: Take ratio for t3/4 and
t1/2for first order reaction and proceed.
Qulem
– 12: A substance S having half life of 100 minutes reacts following
first order kinetics. Calculate the fraction of initial concentration of S,
which will be reacted in 300 minutes.
Solution: We need
{a —
(a — x)} / a = ?
For
first order reaction,
(a — x)
/ a = Exp (— Kt)
Half
life = 100 minutes
=>
K = (ln 2)/100
Thus (a — x) /
a = Exp (— Kt)
=>
(a — x) / a
= Exp [{(—ln
2)/100} 300]
=
2^—3
=
1/8
Hence
fraction of initial concentration reacted = 1 – 1/8 = 7/8
Or
For first order reaction, the amount of substance left after n half lives,
N = N0 (1/2)^n
Since
in the above reaction half life = 100
minutes => 300
minutes = 3 half lives => n = 3
=> N
= N0 (1/2)^3 = N0 / 8
Thus
he required answer, (N0 – N) / N0 = 1 – 1/8 =
7/8
Qulem
– 13: The decomposition of N2O5 (g) into NO2 (g)
and O2 (g) follows first order kinetics, in which the rate
constant is 7.48 X 10-3 S^-1. If initially only N2O5 was
present at 0.1 atm, determine the time needed to raise the pressure to
0.145 atm.
Solution:
The
total pressure = Pt = P0 – 2P + 4P + P = P0 + 3P
=>
2P = (2/3) (Pt – P0)
The
partial pressure of N2O5 = P0 – 2P = P0 – (2/3) (Pt
– P0)
We
are to raise the total pressure to 0.145 atm. Thus when Pt = 0.145 atm,
The
partial pressure of N2O5 = P0 – (2/3) (Pt – P0) =
0.07 atm
Applying
kinetics equation w.r.t. pressure, t = (2.303 /
k) log (P0 / PA)
=>
t = (2.303 / 7.48 X 10-3 S-1) log
(0.1 / 0.07) = 47.7 sec
Some
important first order reaction and their experimental measurement:
1.
Decomposition of Hydrogen peroxide in water: H2O2 decomposes
in water in presence of finely divided Platinum following first order kinetics.
H2O2 —> H2O
+ ½ O2
As the reaction proceeds the concentration of H2O2 decreases
and that of oxygen increases. Accordingly the progress of the reaction can be
measured by titrating equal volume of reaction mixture (withdrawn at regular
intervals) against a standard KMnO4 solution.
Volume of KMnO4 consumed at the beginning of
reaction can be considered as the initial concentration of the H2O2 and
accordingly the volume of KMnO4 at any time t can be considered
to be the remaining unused H2O2.
Thus V0 = The volume of KMnO4 used
at the beginning ≡ Initial
concentration of H2O2 ≡ a
And Vt = The volume of KMnO4 used
at different time interval ≡ Amount of
unused H2O2 ≡ a — X
Qulem – 14: Following is a set of data for the decomposition of H2O2. Show that the reaction is of first order.
|
Time (s) |
0 |
600 |
1200 |
|
KMnO4
(ml) |
22.8 |
13.8 |
8.3 |
Here V0 = a = 22.8 ml and Vt = a — x = at 600 sec = 13.8
and at 1200 sec = 8.3 ml
At 600 sec
K = (2.303 / 600) log (22.8 / 13.8)
For 1200 sec
K = (2.303 / 1200) log (22.8 / 8.3)
Find the value of K in both of these two cases. You will
find K in both of the cases equal or nearly equal which will prove it to be the
first order.
2. Hydrolysis of methyl or ethyl acetate: In this case, the progress of the reaction is measured in
terms of formation of product. The formation of acetic acid is measured by
titrating against standard NaOH solution.
CH3COOR + H2O —> CH3COOH + ROH
In such case, initial concentration of the ester ≡ concentration
of acetic acid measured at end of the reaction — concentration of acetic acid measured initially
≡ V∞ — V0 ≡
a
And V∞ — Vt ≡ a —
X
3. Decomposition of N2O5 in
CCl4:
In this case, the volume of oxygen is measured to
follow the progress of the reaction.
N2O5 —> N2O5 +
½ O2
N2O4 —> 2NO2
In this case, V∞ ≡ Volume
of oxygen collected at the end of reaction (infinite time)
Initial amount of N2O5 ≡ a ≡ V∞ and
Vt ≡ Volume of oxygen collected at time t ≡ X and
thus V∞ — Vt ≡ a —
X
Now we can apply the formula:
K = (2.303 / t) log { V∞/ (V∞ — Vt)}
4. Inversion of cane Sugar: C12H22O11 + H2O —>
C6H12O6 + C6H12O6 .
In such case, the progress of the reaction is studied by measuring the angle of
rotation of the reaction mixture. Sucrose is dextro-rotatory where as the
mixture product is laevo-rotatory. This conversion from d- to l- (positive to
negative angle of rotation) is called inversion of sugar.
r0 — r∞ ≡
a; r0 — rt ≡ X;
rt — r∞ ≡ a —
X
Now we can apply the formula:
K = (2.303 / t) log {(r0 — r∞) / (rt — r∞)}
Qulem – 15: The optical rotation values in the inversion of sucrose using 0.55N HCl at 270C is given below. Find out the order of the reaction.
|
Time
(min) |
0 |
10 |
20 |
40 |
∞ |
|
Rotation
(Degrees) |
+32.4 |
+28.8 |
+25.5 |
+19.6 |
—11.1 |
5. Radioactive Decay: The radioactive disintegrations follow first order kinetics.
Qulem – 16: The half life of C^14 isotope is 5730 years.
An wooden vessel found from certain archeological activity has only 80% of the
C^14 found in a freshly cut wood. Estimate the age of the
sample.
Solution: Take a = 100 and a — X = 80. Calculate K
from half life and put the formula to get answer
nth order reaction: For any reaction of order n, we can derive the integrated
rate equation:
(n — 1) K.t
= {1/At^(n—1)} —
{1/A0^(n—1)} (Except n = 1)
Putting At =
A0 (1 — α) we have,
tα = {1/(A0^(n—1)}
[(1/k(n—1){(1/(1 — α) ^(n—1)) —1}]
Accordingly, for a second order
reaction (n=2), we have,
Kt = (1/At) — (1/A0)
and t1/2 = 1/ A0K
Collision theory of reaction rate: According to collision theory,
Reactants are made up of molecules and are always in a state of random motion.
They go on colliding with one another.
The collision frequency (Z) is the number of intermolecular collisions per unit volume per
second at a given temperature.
Any collision which is effective brings about the chemical reaction. A collision becomes effective when it satisfies the following conditions:
i.The molecules
colliding should possess energy equal to or greater than a certain minimum value of energy known as threshold energy. This energy is related to the activation energy.
ii .They should have proper orientation.
Collision theory can be discussed for
two cases:
1. Lindemann’s
Theory:
Lindemann proved how some reaction
seems to be bimolecular but follow first order kinetics.
Consider a reaction A --------->
Products
Lindemann proposed the following
mechanism:
A+A ---------> A* + A
(Activation) ------- i
A +A* ----------> A + A
(Deactivation) ------- ii
A* -----------> Products
(Reaction) --------- iii
Lindemann explained that there exists a
time lag between the activation and reaction.
In case the time lag is long, then step
iii is slow and the reaction follows first order kinetics but if the time lag
is short, then step ii is slow and the reaction will follow second order
kinetics.
Lindemann also explained how pressure
affects kinetics of reaction.
2. For an elementary
bimolecular reaction:
A + B —> product
If Z AB is the collision frequency and
If ‘f’ is the fraction of collisions between molecules which possess energy equal to or greater than a
certain minimum value of energy known as activation energy,
Then the rate of reaction, r = dx/dt ∝ ZAB x f ……….eq. 1
Further according to kinetic energy of gases,
The fraction of molecules which possess energy equal to or greater than a certain minimum value
of energy known as activation energy, ‘Ea’ at a temperature T is given by Boltzmann factor (f).
f = e^—Ea/RT
Thus substituting for ‘f’ in eqn (1) we get
r = rate ∝ ZAB x e^—Ea/RT
As rate of reaction is directly related to a rate constant ‘K’ thus we can write
K ∝ ZAB x e^—Ea/RT --------- eq. 2
To account for the orientation factor (for making the collision effective) another factor (P)
called orientation factor or probability factor should be introduced.
Hence, the eq (2) becomes K = P. ZAB x e^— Ea/RT
Limitations of collision theory:
1. For some reactions, the calculated and experimental value of K very widely
2. It fails to explain rate of reversible reaction
3. It is not possible to predict the correct orientation before the reaction collide effective
Concept of activation energy and activated complex:
The reactants in a chemical reaction must acquire the threshold energy
to bring about the
product. The extra energy supplied to the reactant to attain the threshold energy and to undergo
the chemical reaction is called the activation energy.
Activation (Ea) = Threshold (Eth) — Average kinetic energy of reactant (E R)
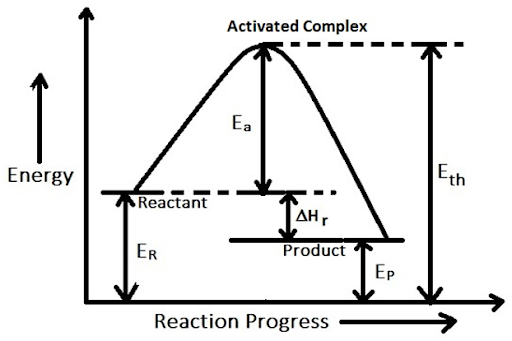
To illustrate the concept of activation
energy, we can take the example of the reaction between methane and oxygen.
They don’t react at ordinary temperature even though they collide with each
other because they posses energy less than threshold energy. But
when a lighted match stick is brought near the reaction mixture they gain
additional energy to reach threshold energy and begin to react. The heat
produced help in continuing the reaction of other molecules.
The value of activation energy decides the fraction of total number of collisions which is
effective. Clearly if the value of activation energy is low, large number of molecule will possess this
energy. Hence the number of effective collisions will be more. So fast reactions have low activation
energy and slow reaction have high activation energy.
For
Example, even though concentration and temperature remains constant, rate of
reaction varies from reaction to reaction. For example,
NO
(g) + ½ O2 (g) ---------> NO2 (g) (Fast)
CO
(g) + ½ O2 (g) ---------> CO2 (g) (Slow)
Since the bond in CO is stronger than NO, the activation energy of CO is greater than NO. Hence the oxidation of CO is slower than that of NO.
Exothermic and endothermic reaction:
Exothermic reaction: In exothermic reaction, energy of product is less than that of reactant.
Thus the enthalpy of the reaction,
ΔH r = ΣHP — ΣHR = — ve . That means energy is lost.
Endothermic reaction: In endothermic reaction, energy of product is greater than that of reactant.
Thus the enthalpy of the reaction,
ΔH r = ΣHP — ΣHR = + ve . That means energy is gained.
Activated complex: An activated complex is an intermediate state that is formed during the
conversion of reactants into products. It is the structure that results in the maximum energy point in the reaction
path. An activated complex may give rise to product or may be deactivated to
the reactant.
Activation energy in reversible reaction:
In reversible reaction,
Activation energy (Ea) for forward reaction = Ea (forward) = Eth — ER
Activation energy (Ea) for backward reaction = Ea (backward) = Eth —
Ep
If Ea (forward) > Ea (backward), then reaction is endothermic.
If Ea (forward) < Ea (backward), then reaction is exothermic.
Effect of temperature on the rate of reaction:
It has been already been explained, for most of the reaction, the rate of reaction comes
nearly double or even more for 10 0c rise in temperature. This is
explained in equation
as temperature co efficient = K (t+10) / K (t0c) = 2 to 3
Explanation:
This can be explained considering the increase in effective collision. The following graph makes it clear,
We know from kinetic theory, kinetic energy is directly proportional to the temperature.
From the above graph of molecular velocity, the point ‘a’ represents the minimum kinetic energy
that should be possessed by molecule for effective collision. The area ‘abcd’ represents the total
number of molecules having energy greater than the threshold value at temperature t0c.
When the temperature is increased to (t + 10) 0c, the curve shifts to the area ‘abef’. Thus the
fraction of molecules having kinetic energy greater than threshold value is now almost double than
the area abcd. Hence the increase in rate of reaction with increase in temperature is mainly due to
the increase in the number of effective collision.
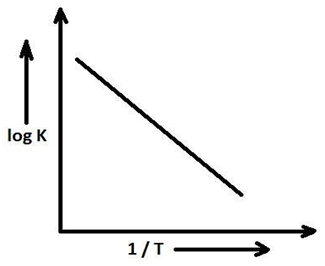
Arrhenius equation:
A quantitative approach which relates the rate of reaction (rate constant),
temperature and activation energy is given by Arrhenius known as Arrhenius equation.
The Arrhenius equation is given by,
K = A e ^— Ea/ RT ……….eq. 1 Where K = Rate constant
A = frequency factor
Ea = Activation energy
R = universal gas constant
T = temperature in Kelvin
Taking logarithm in both sides,
ln K = ln A e ^— Ea/ RT
=> ln K = ln A + ln e ^— Ea/ RT
=> ln K = ln A — Ea/ RT
=> 2.303 log K = 2.303 log A – Ea / RT
=> log K = log A – Ea / 2.303 RT ----------- eq. 2
When a graph is plotted for log K vs ‘1 / T’ we get a straight line where the intercept is ‘log A‘ and the slope is — Ea / 2.303R.
If K1 and K2 are the two rate constants at two different temperatures T1 and T2 respectively, then,
log K1 = log A – Ea / 2.303RT1 -------- eq.3
& log K2 = log A – Ea / 2.303RT2 -------- eq. 4
Subtracting eq (3) FROM (4)
log K2 – log k1 = (Ea / 2.303RT1) – (Ea / 2.303RT2)
log (K2 /K1) = Ea / 2.303R (1/T1 – 1/T2)
log (K2 /K1) = Ea / 2.303R
{(T2 – T1) / T1 T2} --------- eq. 5
The equation (5) is the logarithmic form of Arrhenius equation which gives the variation of rate
constant with temperature. Knowing all other values, activation energy can be calculated.
Qulem
– 17: The rate constants of a reaction at 500c and 1000c
are found to be 1.5 X 107 s-1 and 4.5 X 107 s-1.
Calculate the activation energy.
Solution: Put R = 8.314 j k-1 mol-1 and proceed
To be continued ....
A Quality book is like a best friend.
Let’s enjoy the friendship.










Hi ! Please Do Not Spam in Message and Be Honest and Respectful.